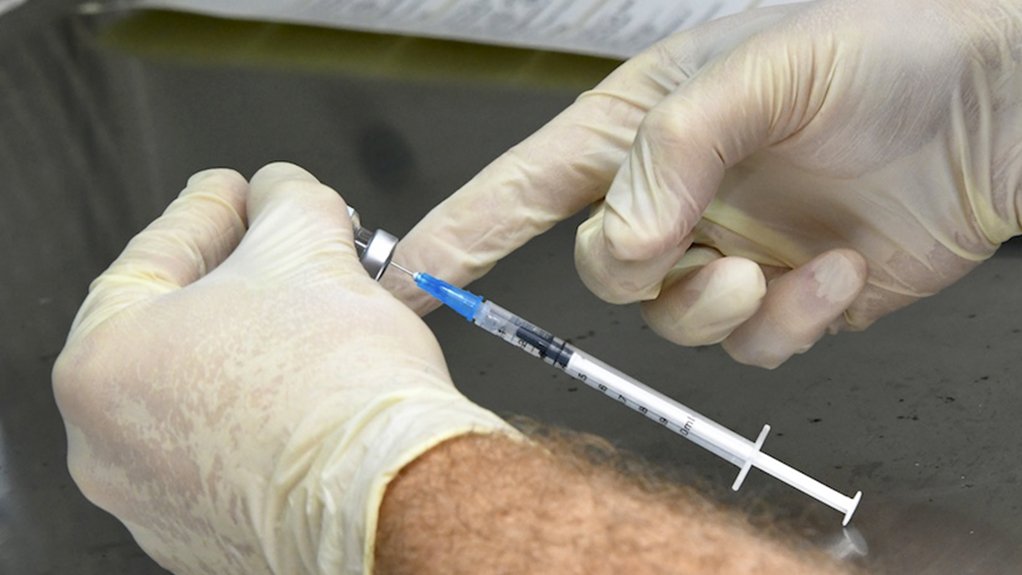
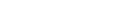The South African Health Products Regulatory Authority (SAHPRA) has said that no major safety concerns with the Johnson and Johnson (J&J) vaccine have been identified so far and it is expecting updates in a few days.
On Tuesday, Health Minister Dr Zweli Mkhize announced the voluntary suspension of the J&J Covid-19 vaccine rollout in South Africa.
The decision was taken following the US Food and Drug Administration’s (FDA's) recommendation to temporarily pause the J&J vaccine programme after six women had developed blood clots after taking the vaccine. The US has vaccinated over 6-million people with the J&J vaccine.
SAHPRA met with the Sisonke study team and the J&J manufacturer Janssen Pharmaceutical on Tuesday.
SAHPRA and scientific expert committees have recently reviewed the data from the Sisonke Phase 3b implementation study and have found no causal relationship between vaccination and the development of blot clots.
SAHPRA requested a pause in the Sisonke Protocol study so that it can review the data.
To date, close to 300 000 South African healthcare workers have taken the J&J vaccine and so far, no rare blood clots have been detected with close monitoring.
Further data are being obtained from the manufacturer and the US FDA.
EMAIL THIS ARTICLE SAVE THIS ARTICLE ARTICLE ENQUIRY
To subscribe email subscriptions@creamermedia.co.za or click here
To advertise email advertising@creamermedia.co.za or click here