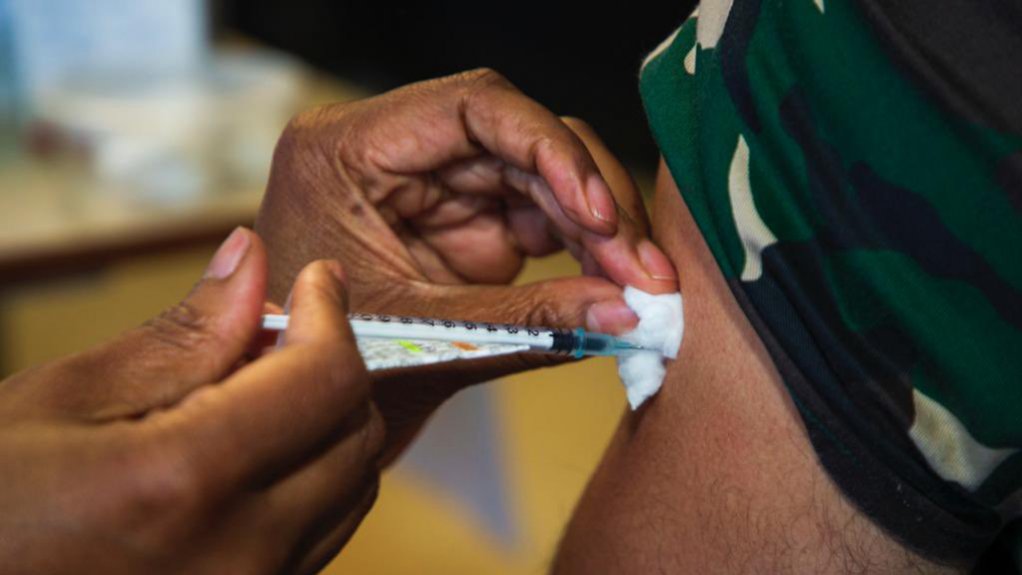
ImmunityBio, the US company controlled by biotech billionaire Patrick Soon-Shiong, is planning to conduct a trial for a second Covid-19 shot in South Africa.
The new vaccine will combine the use of both ribonucleic acid, or RNA, and deoxyribonucleic acid, or DNA, as well as the adenovirus used in a an inoculation that’s already being tested, Soon-Shiong said in an interview.
ImmunityBio is already running local trials of an earlier dose, known as the hAd5 T-cell vaccine, and will test it as a booster for South African healthworkers who have received Johnson & Johnson’s vaccine.
The new programme’s co-ordinators “will likely start enrolling by mid-November, contingent on getting the necessary approvals” from the South African Health Products Regulatory Authority and the trial’s ethics committee, said Shabir Madhi, a vaccinologist at Johannesburg’s University of the Witwatersrand, who will be running the process.
While drugmakers around the world have produced billions of approved Covid-19 vaccines to roll out across the globe, many international pharma companies such as ImmunityBio have turned their attention to second-generation doses that could be used to fight future coronavirus variants and potentially deadly new waves.
South Africa has been the site of a number of trials already, carried out by Pfizer, AstraZeneca, Johnson & Johnson, Novavax and Sinovac Biotech. Madhi ran both the AstraZeneca and Novavax initiatives.
The new vaccine is expected to bolster the production of both antibodies and T Cells that kill virus-infected cells.
“We hope that this then solves the problem of high antibodies and durable immunity,” Soon-Shiong said, adding that the trial will initially encompass the first two phases of the three-stage path to approval for emergency or general use.
At a later stage, ImmunityBio may also run a trial of a so-called subunit protein Covid-19 vaccine, Soon-Shiong said. Subunit vaccines use fragments of viruses to stimulate an immune response.
EMAIL THIS ARTICLE SAVE THIS ARTICLE
To subscribe email subscriptions@creamermedia.co.za or click here
To advertise email advertising@creamermedia.co.za or click here